Palladium-Catalyzed Aminomethylation and Cyclization of Enynol to O-Heterocycle Confined 1,3-Dienes | Organic Letters

Palladium‐Catalyzed Sequential Cyclization/Functionalization of Oxime Ethers with Unactivated Vinyl Ethers for Tunable Assembly of Structurally Diverse Isoxazoles - Li - 2021 - Chinese Journal of Chemistry - Wiley Online Library

Marine furanocembranoids-inspired macrocycles enabled by Pd-catalyzed unactivated C(sp3)-H olefination mediated by donor/donor carbenes | Nature Communications

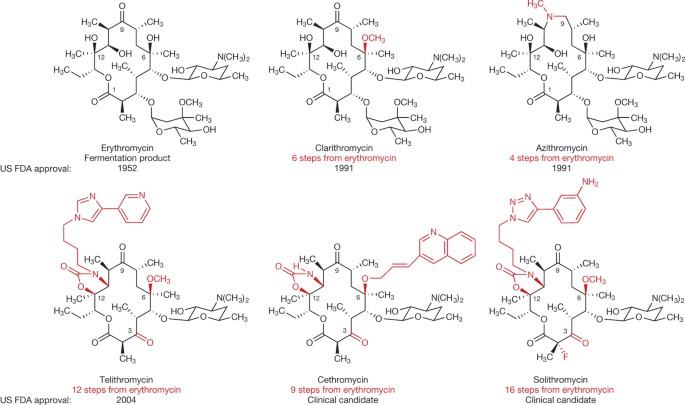
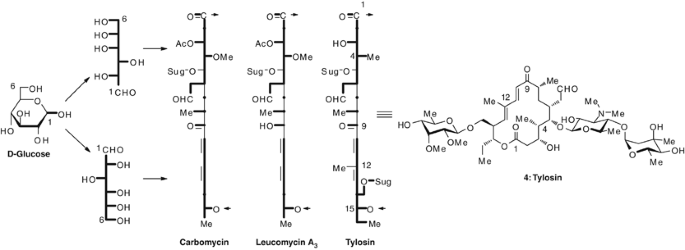
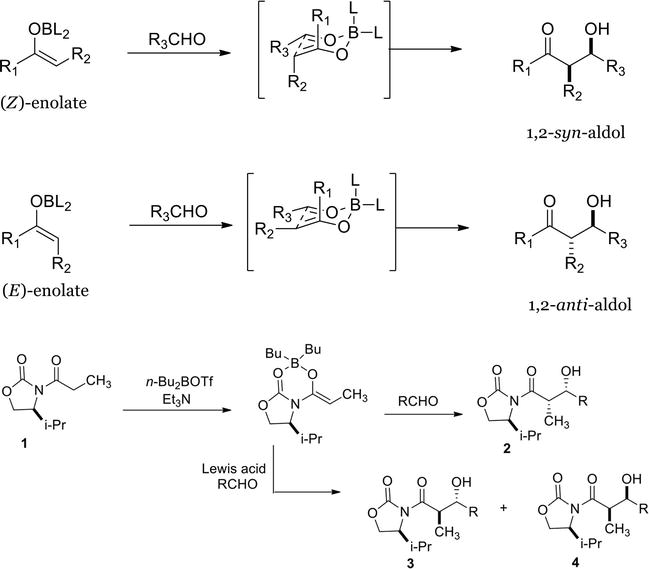
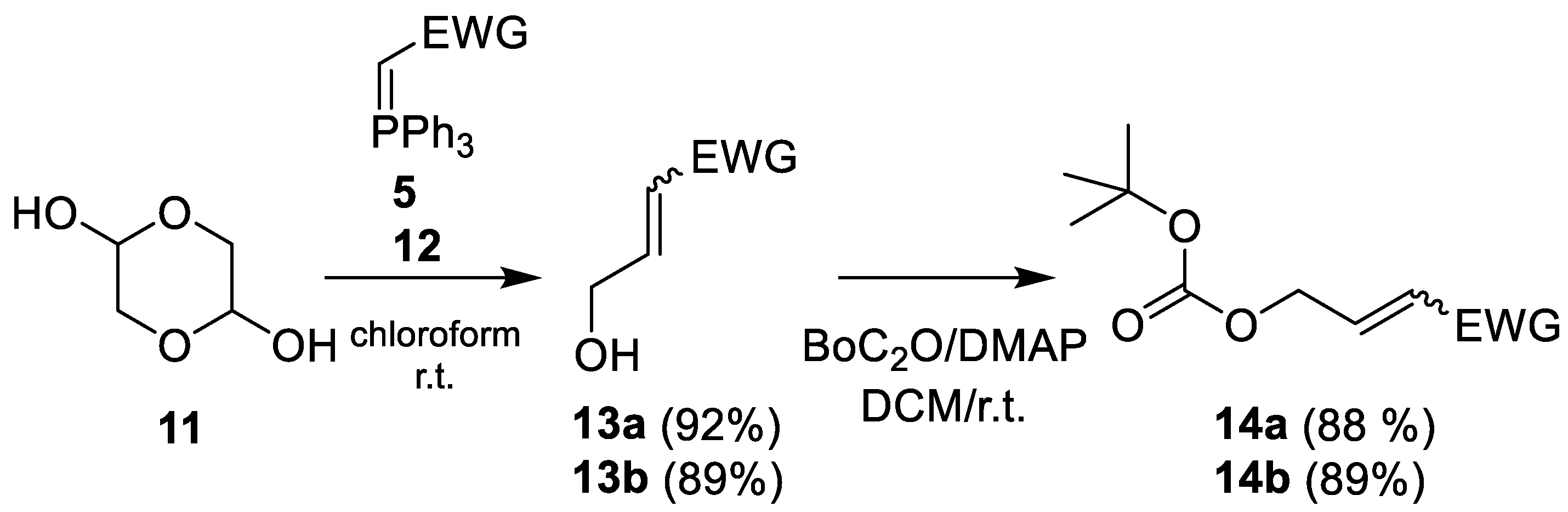
Molecules | Free Full-Text | Scaffold Modifications in Erythromycin Macrolide Antibiotics. A Chemical Minireview

Molecules | Free Full-Text | Scaffold Modifications in Erythromycin Macrolide Antibiotics. A Chemical Minireview

Iron-catalyzed synthesis of N-heterocycles via intermolecular and intramolecular cyclization reactions: A review - ScienceDirect

Tsuji–Trost Cyclization of Disulfonamides: Synthesis of 12-Membered, 11-Membered, and Pyridine-Fused Macrocyclic Triamines | ACS Omega

Nickel-Catalyzed Enantioselective Coupling of Aldehydes and Electron-Deficient 1,3-Dienes Following an Inverse Regiochemical Course | Journal of the American Chemical Society

Operando Spectroscopic and Kinetic Characterization of Aerobic Allylic C–H Acetoxylation Catalyzed by Pd(OAc)2/4,5-Diazafluoren-9-one | Journal of the American Chemical Society
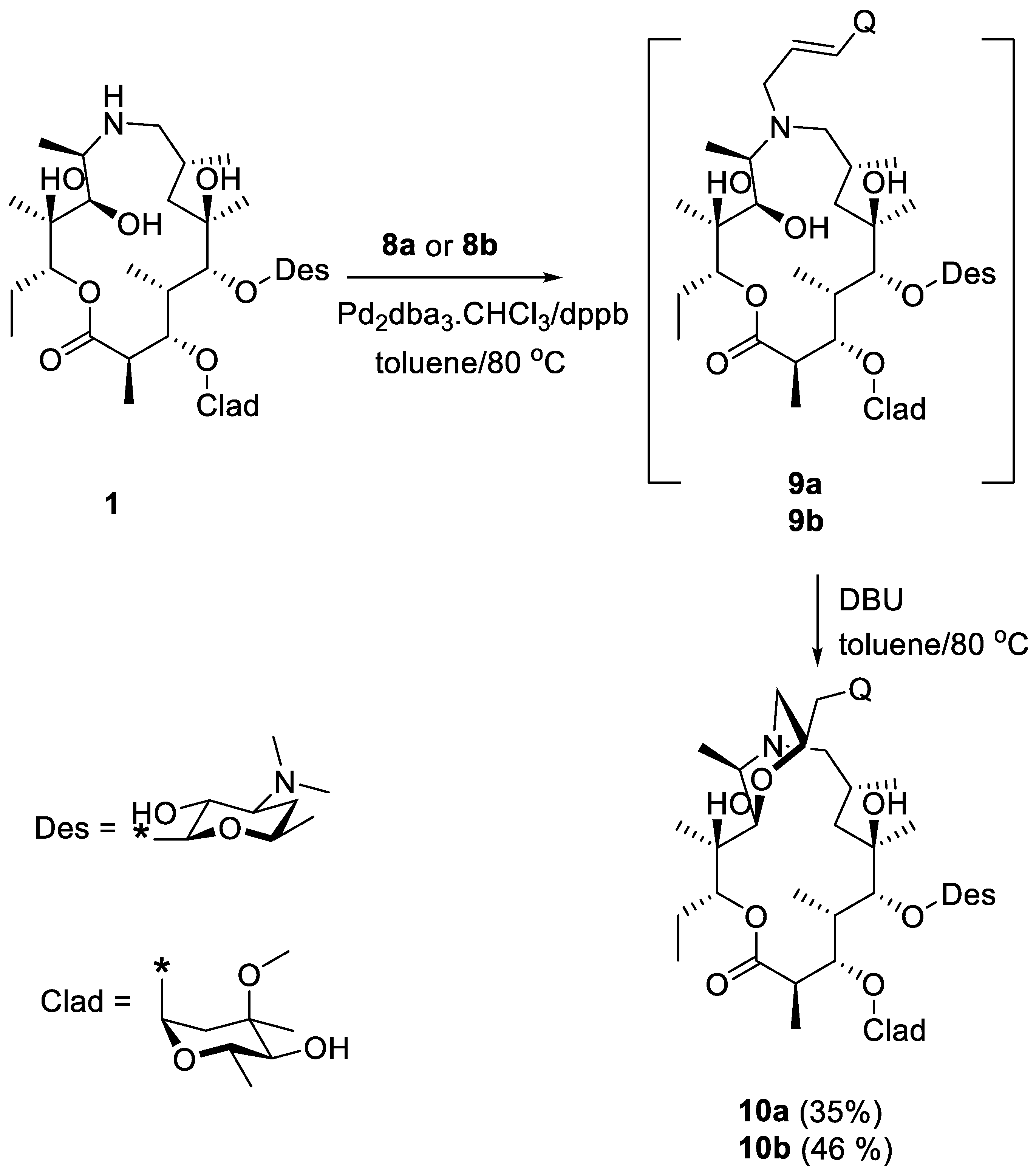
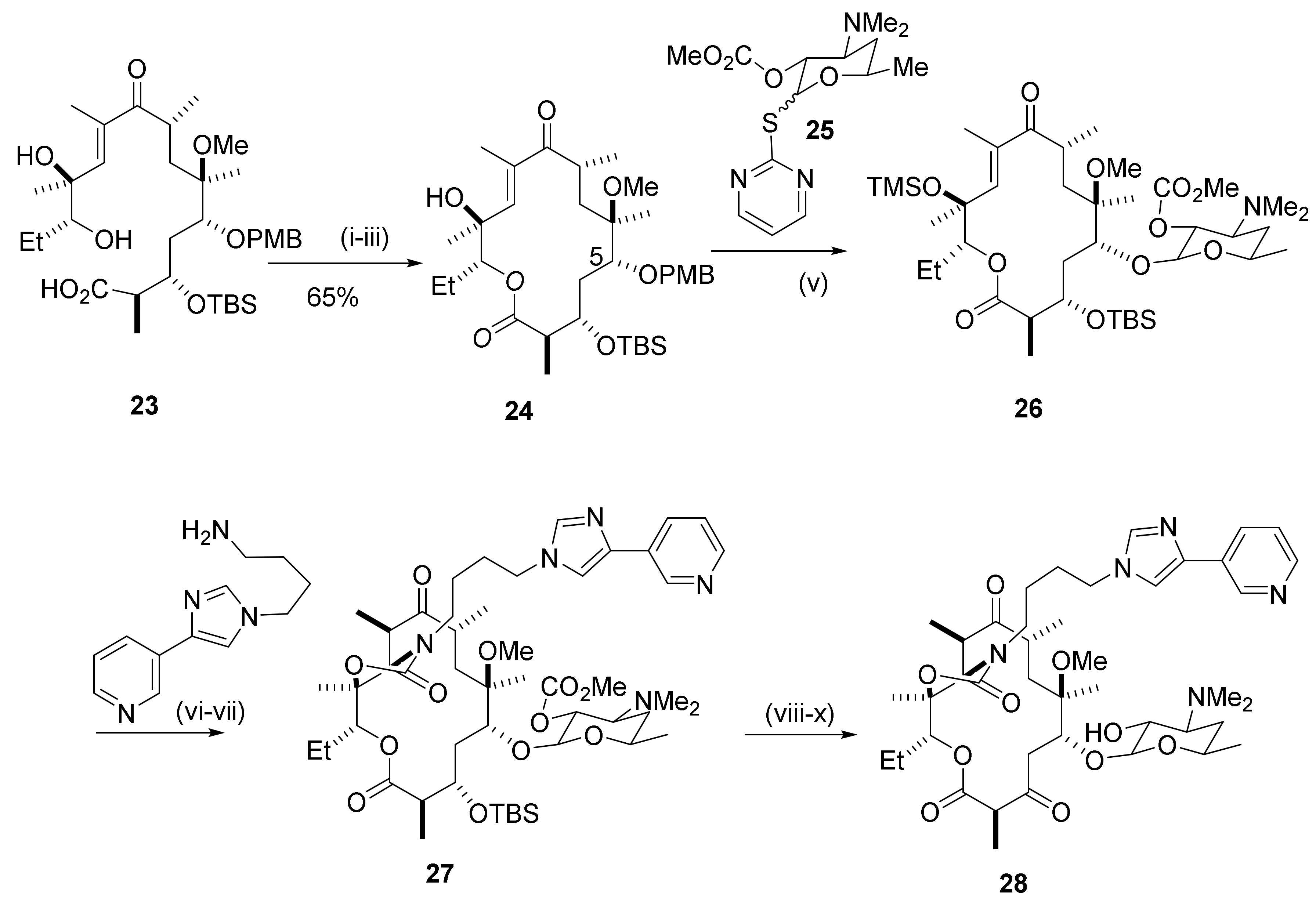
Molecules | Free Full-Text | New Bicyclic Azalide Macrolides Obtained by Tandem Palladium Catalyzed Allylic Alkylation/Conjugated Addition Reaction

Black Swan Events” in Organic Synthesis - Nugent - 2012 - Angewandte Chemie International Edition - Wiley Online Library

Cyclization catalyzed by palladium(0). Initial studies and macrolide formation | Journal of the American Chemical Society

Molecules | Free Full-Text | Scaffold Modifications in Erythromycin Macrolide Antibiotics. A Chemical Minireview
![A Palladium-Catalyzed Cyclization−Oxidation Sequence: Synthesis of Bicyclo[3.1.0]hexanes and Evidence for SN2 C−O Bond Formation | Journal of the American Chemical Society A Palladium-Catalyzed Cyclization−Oxidation Sequence: Synthesis of Bicyclo[3.1.0]hexanes and Evidence for SN2 C−O Bond Formation | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja070919j/asset/images/large/ja070919jh00003.jpeg)
A Palladium-Catalyzed Cyclization−Oxidation Sequence: Synthesis of Bicyclo[3.1.0]hexanes and Evidence for SN2 C−O Bond Formation | Journal of the American Chemical Society
Molecules | Free Full-Text | Scaffold Modifications in Erythromycin Macrolide Antibiotics. A Chemical Minireview

Enantioselective Synthesis of 2,6-cis-Disubstituted Tetrahydropyrans via a Tandem Catalytic Asymmetric Hydrogenation/Oxa-Michael Cyclization: An Efficient Approach to (−)-Centrolobine | Organic Letters
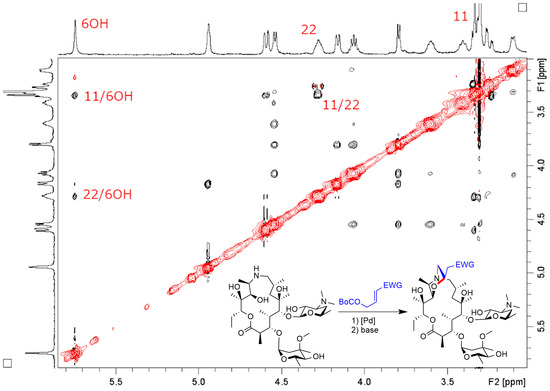
Molecules | Free Full-Text | New Bicyclic Azalide Macrolides Obtained by Tandem Palladium Catalyzed Allylic Alkylation/Conjugated Addition Reaction
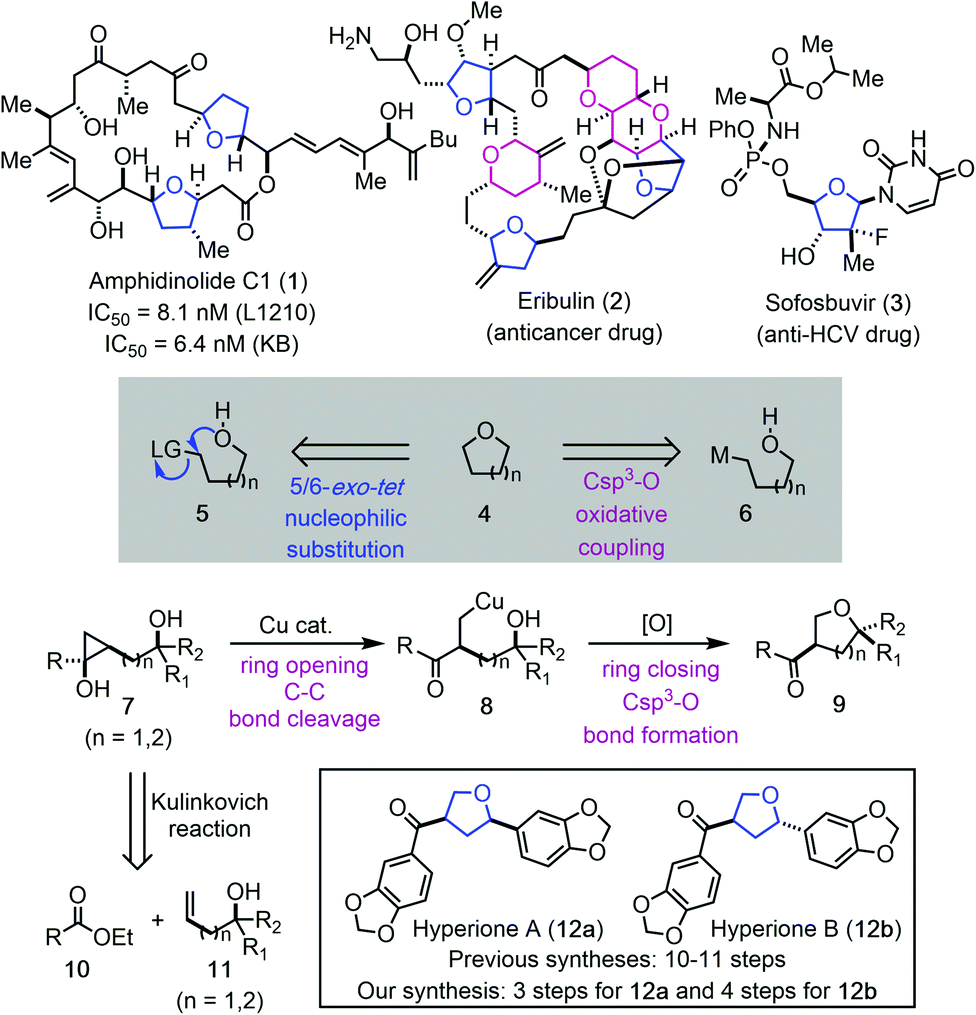
Cu-catalyzed hydroxycyclopropanol ring-opening cyclization to tetrahydrofurans and tetrahydropyrans: short total syntheses of hyperiones - Chemical Science (RSC Publishing) DOI:10.1039/D0SC05556E

Palladium-Catalyzed Stereoselective Cyclization of in Situ Formed Allenyl Hemiacetals: Synthesis of Rosuvastatin and Pitavastatin | Organic Letters